What’s a polymer? We think these might just be one of the most amazing things–that most non-scientists know practically nothing about.
We “ordinary folks” have always assumed polymers are just another kind of plastic that we can blame for the current state of the planet. Clearly, though, we have a lot to learn about these amazing substances and the phenomena by which they’re formed
So, this article will take you along with us on our quasi-scientist journey through the remarkable world of polymers like thermoplastic polymer. You won’t be confused, we promise!.
What’s a Polymer?
A polymer is “a substance which has a molecular structure built up chiefly or completely from a large number of similar units bonded together, e.g. many synthetic organic materials used as plastics and resins.”
Polymers are large molecules formed by bonding (i.e., chemically linking) a series of smaller building blocks. A polymer is a chain, and each of its links is a monomer. Monomers can be simple–maybe a few atoms or maybe dozens.

Polymers, though, are very complex. Sometimes, they are very long chains and sometimes even branching networks. whatever their shape, they can be so huge that they have been classified as macromolecules.
They’re are like building toys, like Legos, that are comprised of identical or very similar pieces you can join together forever–or until you run out of pieces. So, it seems many of us have been thinking about polymers since we were children.
Types of Polymers
You might be surprised to learn that many substances found in nature are polymers. These are called natural polymers. Their structures are both echoed and further developed in the artificial polymers developed in laboratories.
A difference between the two types of polymers is that in artificial polymers, links in the polymer chain are often identical whereas, with natural polymers, there are variations among the linked pieces. We’ll look at both types in this section.
Natural Polymers
Every substance is made from unique chemical bonds that define its physical properties and functions. It can be surprising (and maybe a little disconcerting) to discover that we humans and everything that surrounds us are made from these bonds.
Understanding our world in this way is what the field of chemistry is all about. And using this understanding to develop new substances and products is what chemical engineering is about
Artificial Polymers
The ostensible purpose of artificial polymers is to solve problems or enhance what’s already working. For example, polymers are used in shampoos and hair conditioners to make hair stronger.
A self-described “curly-haired polymer scientist” explains that “cationic polymers are quite are often used as conditioners. They’ve been chemically modified to have positive charges along their backbone…
Since hair is negatively-charged, these polymers become bound electrically to the surface of your hair when applied in the shower and resist being rinsed off by the water.”
This is just one example of how polymers make everyday products more effective. We’ll discuss this in the next section.
What Other Kinds of Things Are Made From Polymers?

This question opens a floodgate of information. There is virtually no end to the number and types of polymers in the world–including those yet to be developed. We’ll go through each type.
Natural Polymers
It’s startling to find out how polymers are so integral to our existence–as body parts, food, and manufactured materials and items we use every day. Here’s a partial list:
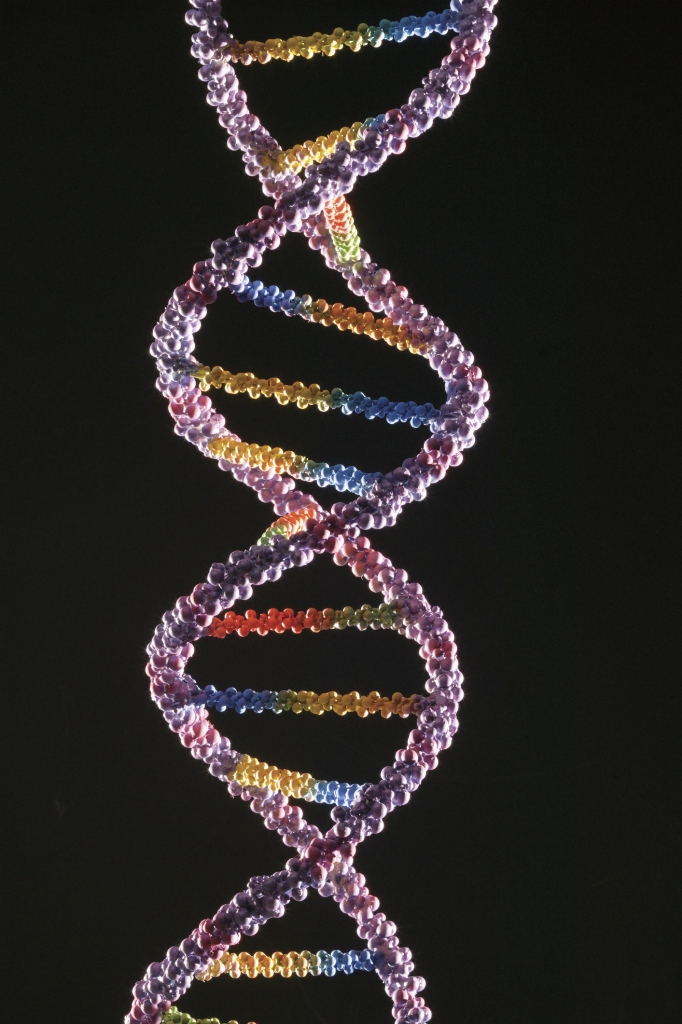
RNA and DNA: The Building Blocks of Life
The following shows how two vitally important polymers function in living organisms.
Ribonucleic acid (RNA) and deoxyribonucleic acid (DNA) are polysaccharides. Their polymer backbones are based on sugar units. It’s molecular groups attached to these units that give them their unique properties, though.
DNA and RNA differ based on the presence or absence of a single oxygen atom. They also differ because DNA is a two-strand molecule while RNA is only one.
DNA is responsible for storing and transferring genetic information. RNA directly codes for amino acids and acts as a messenger between DNA and ribosomes to make proteins.
Scientists have gathered a great deal of evidence that RNA was the “original molecule of life.” This is because of its ability to both store hereditary information and provide functional activity as an enzyme.
Other Polysaccharides
Other polysaccharides include wood (in the cellulose family) and vegetables (like corn and potatoes) that are in the starch family.
Here’s an example to demonstrate how much the properties of natural polymers can differ. Starches are highly soluble and readily dissolve in water, whereas cellulose is very rigid (crystalline) and not very soluble at all.
Chitin
Chitin is a very unique substance. It’s what forms the hard shells of crustaceans and insects. Scientists would love to be able to figure out how to replicate this in a lab since it would seem to have many practical uses.
Other natural polymers include proteins and enzymes, silk, nylon, and rubber. we’d love to discuss all these and others as well, but we need to move on to artificial polymers now.
Artificial Polymers
Along with the hair care products discussed above, chemical engineers have created many new substances and products. Here are a few examples:
Polyurethane coatings protect materials from corrosion, abrasion, weather, etc.
- Polyester is a versatile fabric that is easily dyed and resistant to shrinkage.
- Polyvinyl chloride (PVC), a substance used to produce plumbing pipes. It can be rigid or flexible.
- Kevlar is a very strong, heat-resistant aramid. It’s best known for its use in bullet-proof vests.
- Polypropylene is known for sanitary uses such as piping for potable water and disposable medical containers.
- Neoprene is a family of synthetic rubbers that maintain flexibility in a wide range of temperatures.
Again, we’ve left out far more than what we’ve included here. But a quick web search will turn up many more polymers and information about their uses.
If all this fascinates you (as it does us), here’s a topic you should be interested in: the expanding or shrinking of 3-dimensionally molded parts.
We’ve heard a lot about this lately. If you want to know more, shop here.
From DNA to Artificial Rubber and Beyond: What’s a Polymer?
We hope this article has given you some food for thought.
The building blocks of life are strikingly close to those of the objects and materials that will build our future. Thus, we must think about the mission of science and experimentation. and give credit where it’s due.
There’s no doubt there are dangers of overusing plastics and various
chemicals. However, whole new generations of scientists and engineers–very aware of environmental concerns–now are working to reverse the damage done.
Let’s hope you continue to seek news about them through the fine blogs and publications we produce.